JingRu Li, PhD; Farah Mirza, BDS, MS; Pin-Wen Wang, PhD; Krista Argosino; Kimberly Milleman, RDH, MS, PhD; Jeffery Milleman, DDS, MPA; and Marilyn Ward, DDS
Abstract: The study objective was to evaluate the effect of different interdental oral cleaning modalities on gingivitis and plaque following a 6-week period of home use. This was a randomized, parallel, examiner-blinded study. Study subjects were routine manual toothbrush (MTB) users with moderate to severe gingivitis, aged 18 to 65 years. Subjects were required to have a gingival bleeding score of ≥1 on at least 50 gingival sites per the Gingival Bleeding Index (GBI) and to have a minimum average plaque score of ≥0.6 per the Rustogi Modified Navy Plaque Index (RMNPI) following a 3- to 6-hour plaque accumulation period. Subjects were randomly assigned to one of four groups based on the oral care cleaning modality: (1) NON group: MTB alone, (2) FLS group: MTB plus string floss, (3) MPF group: MTB plus a Philips® Sonicare® Power Flosser with the Quad Stream nozzle, or (4) PPF group: Philips Sonicare power toothbrush plus the power flosser. Safety and efficacy measures (Modified Gingival Index [MGI], GBI, and RMNPI) were assessed at baseline, 2 weeks, and 6 weeks. The primary efficacy endpoint was the reduction in gingival inflammation from baseline to week 6 as measured by the MGI. A total of 260 subjects were randomized and 256 subjects completed the study. The adjusted mean percent reduction in gingival inflammation from baseline to week 6 was 14.90% for the NON group, 13.16% for the FLS group, 33.51% for the MPF group, and 49.30% for the PPF group. Pairwise comparisons indicated that both the PPF and MPF groups were statistically significantly different from both the NON and FLS groups. In conclusion, use of either the Philips Sonicare power toothbrush with the Philips Sonicare Power Flosser or an MTB with the Philips Sonicare Power Flosser was statistically superior to an MTB alone and an MTB used with string floss in reducing gingival inflammation following 6 weeks of home use.
The removal of dental plaque biofilm and food debris is an important step in maintaining a healthy dentition. Dental plaque contains a diverse bacterial flora that adheres to the tooth surface and, if left undisturbed, may continue to accumulate.1,2 This accumulation can cause caries and gingivitis, which can lead to periodontal disease.3 Routinely performing good oral hygiene is perhaps the most important prevention strategy to control these oral pathologies.
The standard oral hygiene regimen utilizes a toothbrush to remove dental plaque from tooth surfaces. However, the human dentition is geometrically complex, which makes it difficult for a toothbrush's bristles to reach all surfaces of the tooth. The buildup of plaque on interdental surfaces can lead to gingivitis and periodontitis. As an oral care regimen, the combination of toothbrushing and interdental cleaning improves oral health outcomes.4 Consumer options for interdental cleaning include string floss, interdental brushes, and powered flossers using a pressurized stream of water. The combination of toothbrushing and water powered flossers has demonstrated better dental plaque and gingival inflammation reduction than manual toothbrushing alone.5
Dental professionals often recommend a comprehensive oral care regimen that includes both toothbrushing and interdental cleaning. Toothbrushing recommendations may vary based on whether they involve a manual toothbrush (MTB) or powered toothbrush (PTB), and, if powered, the type of bristle motion. PTBs with high frequency, high amplitude bristle motion have been shown to be highly effective in removing plaque and reducing gingival inflammation.6,7 The current study compares gingival health and plaque removal efficacy of the MTB alone, the MTB used with string floss, the MTB used with a Philips® Sonicare® Power Flosser (Figure 1), and a PTB used with a Philips Sonicare Power Flosser.
Materials and Methods
This was a prospectively planned, randomized, parallel, examiner-blinded study that was reviewed and approved by an accredited Institutional Review Board (IntegReview IRB, Austin, Texas). The study's primary objectives were to evaluate the effect of different oral cleaning modalities on gingivitis, as evaluated by the Modified Gingival Index (MGI),8 and safety following a period of 6 weeks of home use. Secondary objectives included an additional gingivitis comparison at 2 weeks and comparisons between the oral cleaning modalities on plaque removal and gingival bleeding at both 2 weeks and 6 weeks. Power analysis for this protocol was based on the primary endpoint of reduction from baseline to week 6 in MGI, comparing different cleaning modalities. A sample size of 61 subjects per treatment group was estimated to provide 85% power to detect a difference in reduction in MGI as small as 0.2 using a two-sided t-test after adjusting for multiple comparisons to maintain an overall type I error rate of 0.05 (Hochberg-Bonferroni multiple comparison).
The three efficacy endpoint measures used in this study were the MGI, the Gingival Bleeding Index (GBI),9 and the Rustogi Modified Navy Plaque Index (RMNPI).10 The MGI and GBI metrics were performed on all evaluable teeth at six sites per tooth, and the RMNPI was recorded at 18 sites per evaluable tooth. The safety endpoints were the adverse changes in the oral cavity upon oral examinations and/or subject interview that were "possibly related" or "related" to the test products, adverse events (AEs) that occurred in/around the oral cavity, and any events that were unanticipated adverse device effects.
Subject recruitment included generally healthy individuals that were aged 18 to 65 years, nonsmokers, and regular manual toothbrush users. Subjects were required to have a minimum of 20 scorable teeth (excluding third molars), a minimum average overall RMNPI score of ≥0.6 following a 3- to 6-hour plaque accumulation period, and a gingival bleeding score of ≥1 on at least 50 gingival sites per the GBI. Subjects were excluded if they were regular power toothbrush users or regular users of dental floss. Subjects were provided with a compliance diary to record each interdental cleaning and brushing encounter.
Study participants were randomized to one of the treatment groups according to a predefined randomization schedule and stratified by gender. The four treatment groups for the 6-week home use period were: (1) NON group: MTB alone with no interdental cleaning modality, (2) FLS group: MTB plus string floss (unflavored waxed), (3) MPF group: MTB plus the Philips Sonicare Power Flosser, or (4) PPF group: PTB (Philips Sonicare ExpertClean used in the clean mode and high intensity with Premium Plaque Control brush head) plus the Philips Sonicare Power Flosser. For both the MPF and PPF groups, the power flosser was used in the clean mode with intensity setting 8 and the Quad Stream nozzle.
All subjects were provided with standard fluoride-containing dentifrice to use for the duration of the study and were prohibited from using any other at-home oral hygiene treatments. Subjects using the MTB were instructed to brush at home for 1 minute twice daily for the 6-week period. Subjects using the PTB were instructed to brush at home for 2 minutes twice daily. For the FLS, MPF, and PPF groups, subjects were instructed to use the interdental cleaning modality once daily in the evening per provided instructions. At each study visit, subjects were required to present with 3 to 6 hours of plaque accumulation.
Randomization and subject instruction were performed by unblinded study personnel who did not perform evaluations or assessments related to study endpoints. Efficacy outcomes included the observed response, the reduction from baseline, and the percent reduction from baseline. An analysis of covariance (ANCOVA) model, with baseline score as the covariate, was used to assess treatment response at each visit and compare the response of the PPF and MPF groups to the FLS and NON groups. All statistical tests were two-sided.
Results
In total, 260 subjects were enrolled and randomized to one of the four treatment groups, with 256 subjects completing the study. Treatment groups were well-balanced for age, gender, race, and ethnicity (P > .05). Four subjects terminated the study early (three withdrew, one lost to follow-up).
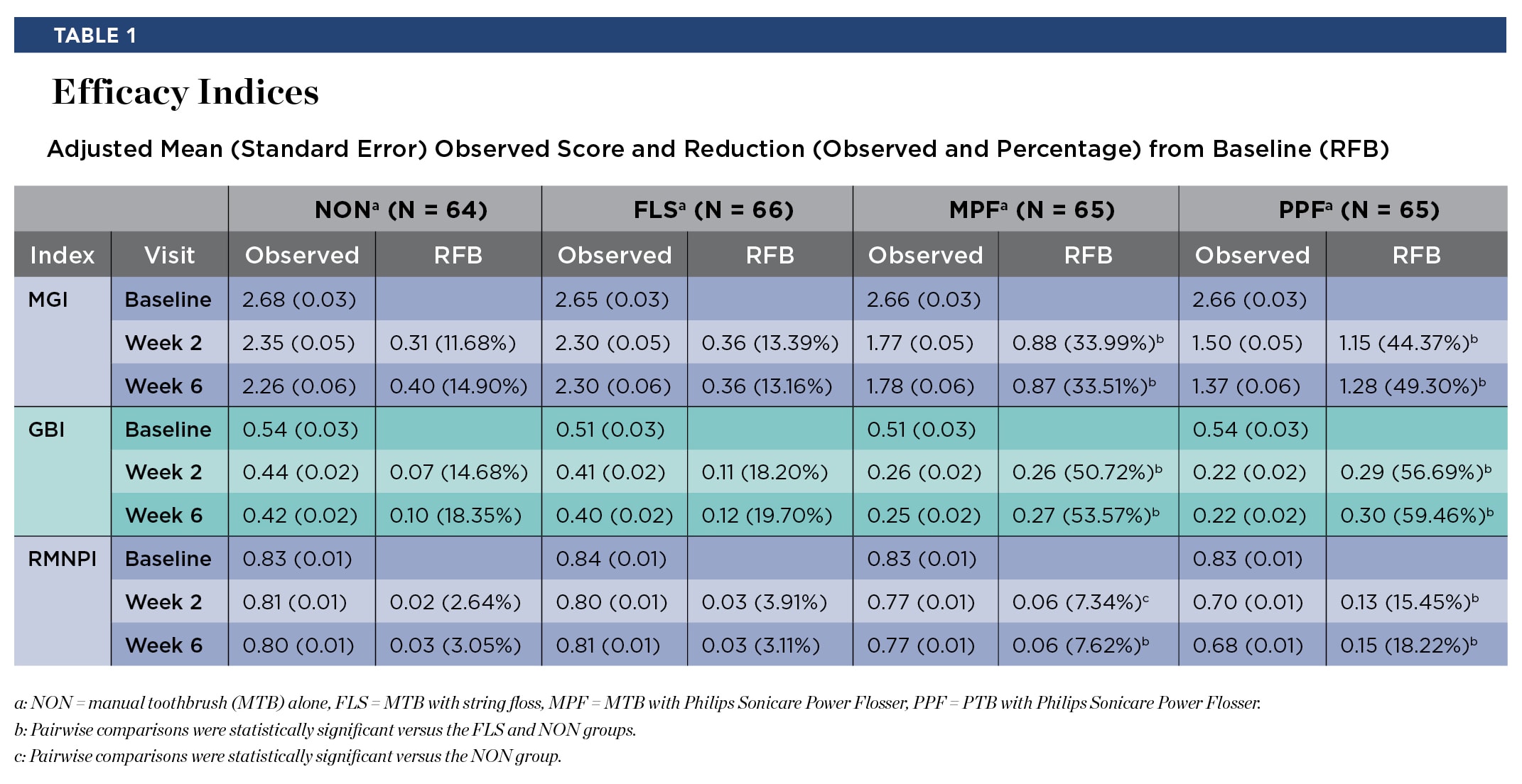
Results for the efficacy scores for all treatments at baseline, week 2, and week 6 can be found in Table 1 for the MGI, GBI, and RMNPI. There were no significant differences between treatment groups in the scores at baseline (P > .05) for any of these indices. In addition to Table 1, the adjusted mean percent reduction from baseline in the three indices scores are presented graphically (Figure 2).
The PPF group was statistically superior to the FLS group (P < .0001) and the NON group (P < .0001) for reduction in MGI, GBI, and RMNPI at both week 2 and week 6. At week 6, the improvement for PPF compared to FLS was 3.6 times higher for MGI, 2.5 times higher for GBI, and 5.7 times higher for RMNPI. The improvement for PPF compared to NON was 3.2 times higher for MGI, 3.1 times higher for GBI, and 5.5 times higher for RMNPI.
The MPF group was statistically superior to the FLS group (P < .0001) and the NON group (P < .0001) for reduction in MGI and GBI at both week 2 and week 6. For RMNPI, MPF was statistically superior to the NON group (P < .04) at both week 2 and week 6; and MPF was superior to the FLS group (P < .04) at week 6, but not week 2 (P = .20). At week 6, the improvement for MPF compared to FLS was 2.4 times higher for MGI, 2.3 times higher for GBI, and 2.4 times higher for RMNPI. At week 6, the improvement for MPF compared to NON was 2.2 times higher for MGI, 2.8 times higher for GBI, and 2.3 times higher for RMNPI.
Three AEs were reported during the study: one in each of the MPF, NON, and FLS groups. The AEs were mild to moderate in severity. The investigator classified two AEs as unrelated to the study and one abrasion along the cervical mucosa in the MPF group as "related" to the study or the study products.
Discussion and Conclusions
The current study investigated whether a Philips Sonicare Power Flosser used once daily unsupervised after either manual or powered toothbrushing improved oral hygiene in subjects over a 6-week period of home use. The results showed that usage of the Philips Sonicare Power Flosser operated in the clean mode at an intensity setting of 8 along with a Quad Stream nozzle is safe and imparts a clinically measurable impact on gingival health status and plaque removal of subjects with moderate to severe gingivitis. Use of the Philips Sonicare Power Flosser, following either manual or powered toothbrushing, resulted in statistically significant gingival health improvements when compared to manual toothbrushing alone or manual toothbrushing with the use of string floss.
Although toothbrushing is commonly used to remove dental plaque from teeth, the interdental space is often neglected as toothbrush bristles are generally not physically able to reach all the interdental surfaces. The power flosser category aims to enhance interdental plaque removal with the convenience and access afforded by a pressurized stream of water. String floss is often recommended by dental professionals and the American Dental Association, and if used correctly and consistently, can provide meaningful interdental cleaning. However, the proper use of string floss is a difficult daily task that requires a significant level of manual dexterity. In a systematic review conducted by Worthington et al, a very low certainty of evidence was found for the efficacy of string flossing as an adjunct to toothbrushing to reduce gingivitis over a 1- to 6-month timeframe.11 In the current study, both the MTB alone (NON) and the MTB plus string floss (FLS) treatment groups confirmed the findings from the Cochrane review that string flossing fails to prevent plaque buildup and reduce gingivitis.11
In conclusion, oral healthcare providers are constantly faced with the task of providing data-driven evidence to recommend treatment regimens for their patients. The results from this randomized human clinical trial can assist dental professionals (both dentists and hygienists) to determine the proper daily homecare regimen for cleaner teeth and healthier gums. Thus, the adjunctive use of the Philips Sonicare Power Flosser, with either a manual toothbrush or the Philips Sonicare power toothbrush, was shown to provide statistically significant oral care benefits through the reduction of gingival inflammation, gingival bleeding, and plaque, compared to manual toothbrushing alone or manual toothbrush plus string floss, following 6 weeks of home use. All products used in the study were safe for home use.
Acknowledgment/Disclosure
The authors thank Jessica Foster, Clinical Operations Manager, Philips Oral Healthcare, for her help with this study. This study was sponsored by Philips Oral Healthcare.
About the Authors
JingRu Li, PhD
Senior Clinical Development Scientist, Philips Oral Healthcare, Bothell, Washington
Farah Mirza, BDS, MS
Senior Clinical Development Scientist and Medical-Dental Safety Officer, Philips Oral Healthcare, Bothell, Washington
Pin-Wen Wang, PhD
Principal Biostatistician, Clinical and Medical Affairs, Philips Oral Healthcare, Bothell, Washington
Krista Argosino
Senior Clinical Research Associate, Philips Oral Healthcare, Bothell, Washington
Kimberly Milleman, RDH, MS, PhD
Director, Compliance Specialist, and Gold Standard Examiner, Salus Research, Inc., Fort Wayne, Indiana
Jeffery Milleman, DDS, MPA
Director, Clinical Operations, and Principal Investigator, Salus Research, Inc., Fort Wayne, Indiana
Marilyn Ward, DDS
Clinical Development Director, Philips Oral Healthcare, Bothell, Washington
References
1. Jakubovics NS, Goodman SD, Mashburn-Warren L, et al. The dental plaque biofilm matrix. Periodontol 2000. 2021;86(1):32-56.
2. Seneviratne CJ, Zhang CF, Samaranayake LP. Dental plaque biofilm in oral health and disease. Chin J Dent Res. 2011;14(2):87-94.
3. Page RC. Current understanding of the aetiology and progression of periodontal disease. Int Dent J. 1986;36(3):153-161.
4. Gallie A. Home use of interdental cleaning devices and toothbrushing and their role in disease prevention. Evid Based Dent. 2019;20(4):103-104.
5. Ren X, He J, Cheng R, et al. The efficacy and safety of oral irrigator on the control of dental plaque and gingivitis: a randomized, single-blind, parallel-group clinical trial. Int J Environ Res Public Health. 2023;20(4):3726.
6. de Jager M, Rmaile A, Darch O, Bikker JW. The effectiveness of manual versus high-frequency, high-amplitude sonic powered toothbrushes for oral health: a meta-analysis. J Clin Dent. 2017;28(1 spec no A):A13-A28.
7. Starke EM, Ward M, Olson M, et al. A randomized parallel study to compare the effects of powered and manual tooth brushing on gingival health and plaque. J Clin Dent. 2019;30(spec no A):A16-A23.
8. Lobene RR, Weatherford T, Ross NM, et al. A modified gingival index for use in clinical trials. Clin Prev Dent. 1986;8(1):3-6.
9. Van der Weijden GA, Timmerman MF, Nijboer A, et al. Comparison of different approaches to assess bleeding on probing as indicators of gingivitis. J Clin Periodontol. 1994;21(9):589-594.
10. Rustogi KN, Curtis JP, Volpe AR, et al. Refinement of the Modified Navy Plaque Index to increase plaque scoring efficiency in gumline and interproximal tooth areas. J Clin Dent. 1992;3(suppl C):C9-C12.
11. Worthington HV, MacDonald L, Pericic TP, et al. Home use of interdental cleaning devices, in addition to toothbrushing, for preventing and controlling periodontal diseases and dental caries. Cochrane Database Syst Rev. 2019;4(4):CD012018.