ABSTRACT
This case report highlights the use of an in-situ hardening alloplastic bone grafting material composed of beta-tricalcium phosphate (β-TCP) granules coated with poly(lactic-co-glycolic acid) (PLGA) to preserve the dimensions and architecture of the alveolar ridge after atraumatic extraction. This material provided a stable scaffold that, although left uncovered, deterred the ingrowth of unwanted soft tissue, allowing newly formed keratinized soft tissue to proliferate over the healing grafted socket and gradually cover the site. At re-entry after 4 months adequate newly formed bone was observed, allowing for the correct positional placement of an implant. The results of this case suggest that an in-situ hardening alloplastic grafting material can be successfully utilized with minimally invasive procedures to preserve the bone and the soft-tissue profile of the alveolar ridge for future implant rehabilitation.
Clinical and experimental studies have shown that tooth extraction leads to bone resorption and atrophy of the alveolar ridge, especially when associated with disease, which may complicate proper future implant placement. Atraumatic extraction followed by immediate socket grafting seems to be a predictable way to preserve the dimensions, contour, and architecture of the alveolar frame and the residual ridge.1-3 Surgical methods that do not require primary soft-tissue closure by flap mobilization have the added benefit of further minimizing patient discomfort and morbidity, while allowing for the preservation of the soft-tissue profile for optimum esthetics and greater predictability.4
In clinical practice several bone graft substitutes of biologic or synthetic origin are being used for socket preservation and bone regeneration prior to implant placement. These materials may vary in composition, mechanical characteristics, and biological mechanism of function regarding resorption and new bone formation, each having its own advantages and disadvantages.1,2
Alloplasts represent a group of synthetic osteoconductive, biocompatible bone substitutes that are free of any risk of transmitting infections or diseases by themselves, and their availability is unlimited. One of the most promising groups of alloplastic bone substitutes are calcium phosphate ceramics, and among them β-TCP is very commonly used.5 Coating the alloplastic graft granules with PLGA can enhance the handling properties and biomechanical characteristics of the material, and produce an in-situ hardening, stable, and at the same time porous and osteoconductive bone graft substitute.6
Case Report
A 65-year-old male patient, non-smoker, without medical contraindication for implant therapy presented with a fractured mandibular right lateral incisor (Figure 1). After thorough examination a delayed implant placement protocol was decided.
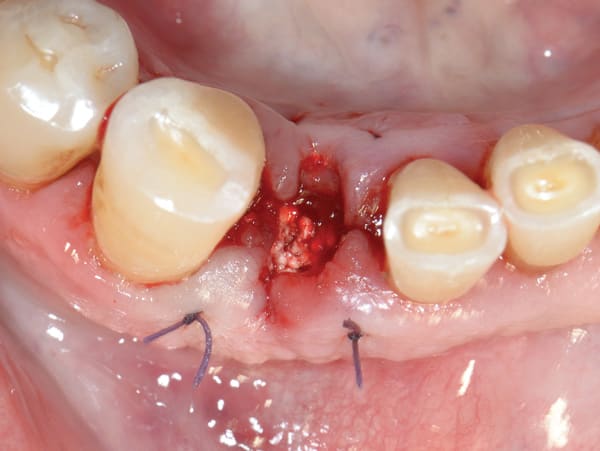
After administration of local anesthesia the fractured tooth was atraumatically extracted using periotomes without raising a flap. Care was given not to damage the surrounding soft and hard tissues and especially the thin buccal bone plate that was identified intact after removal of the tooth fragments. After thorough debridement and rinsing with sterile saline, an alloplastic in-situ hardening bone substitute (GUIDOR® easy-graft® CLASSIC, Sunstar, www.GUIDOR.com) was used to graft the site (Figure 2). It consists of β-TCP granules, which are coated with PLGA. The granules are mixed in a syringe with the provided BioLinker® (N-methyl-2-pyrrolidone solution) (Sunstar). Upon contact with blood or saliva the graft granules adhere to each other forming a sticky, easy-to-handle, moldable mass that begins to harden. Wet gauze can be used to compact the material and accelerate this process so as to form a hard, osteoconductive, porous scaffold for the host osseous regeneration.
The grafted socket was left uncovered in order to heal by secondary intention. A prefabricated provisional removable one-tooth bridge was placed after the surgery without applying pressure to the grafted site.
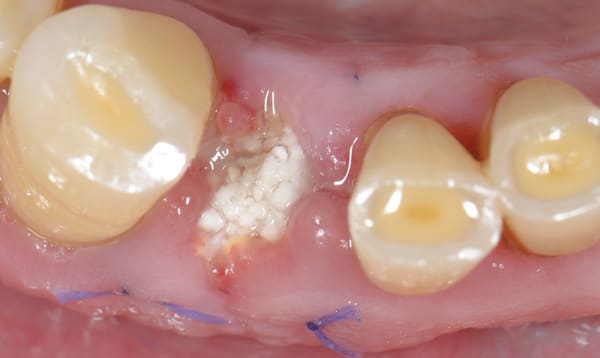
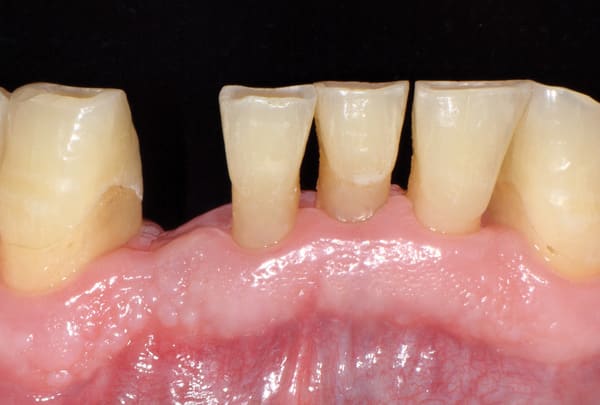
The postoperative healing was uneventful. The biomechanical characteristics of the grafting material permitted the gradual proliferation of the epithelium over the grafted site (Figure 3 and Figure 4), and after 4 months the area was covered with newly formed keratinized epithelium. At that time point clinical examination showed that the volume and architecture of the ridge were adequately preserved (Figure 5).
At re-entry after 4 months, the post-extraction site was filled with newly formed bone. Residual granules were visible, embedded, and in continuity with the regenerated hard tissue (Figure 6). An implant (Paltop Advanced Dental Solutions Ltd., www.paltopdental.com) 3.25 mm in diameter and 11.5 mm in length was inserted at the optimal position (Figure 7), achieving good initial stability (final seating torque: 50 Ncm). The final titanium abutment was placed and a provisional acrylic restoration with a non-functional occlusion was temporarily cemented (Figure 8).
After allowing the soft tissue to mature for 3 months the abutment-level impression was taken and a final metal-ceramic restoration was fabricated (Figure 9). The final clinical outcome was esthetically successful. The radiological examination from the initial situation to the final outcome demonstrated that the socket grafting and the subsequent bone regeneration of the site had been successful regarding biological and functional parameters (Figure 10).
Discussion
In this case a minimally invasive, flapless protocol was followed. No membrane was used to cover the grafted post-extraction site. The mechanical stability of the site, provided by the unique biomechanical properties of the in-situ hardening bone graft substitute, allowed the site to heal by secondary intention.6,7 This resulted in the preservation of the attached keratinized gingiva buccally and permitted the development of newly formed keratinized soft tissue over the grafted site. Primary flap closure patients generally experience more discomfort and the mucogingival junction is significantly more coronally displaced, which may result in esthetic problems and could negatively influence peri-implant soft-tissue health and long-term stability.4,8
The use of β-TCP as a resorbable socket grafting material can preserve space for bone formation until new bone is formed, obtaining high-quality regenerated vital bone, without any nonresorbable grafting material embedded in the bone matrix after several months.9 Thus, the peri-implant bone will be able to adapt according to Wolff’s law after the occlusal loading of the implant through functional remodeling. It is suggested that the long-term presence of residual nonresorbable or slowly resorbable graft particles might interfere with normal bone healing and remodeling, may reduce the bone-to-implant contacts, and have a negative effect on the overall quality and architecture of the bone that surrounds the implant.10
Conclusion
In the presented case, an in-situ hardening alloplastic bone grafting substitute was used in a minimally invasive, successful, and predictable way for socket preservation, resulting in pronounced regeneration of bone capable of supporting implant placement after a 4-month healing period, and capable of remodeling and maturing after loading. The in-situ hardening property of this material may enable clinicians to utilize a flapless procedure without primary wound closure that reduces patient morbidity, preserving the attached keratinized gingiva and allowing for further production of newly formed keratinized soft tissue. Thus, both hard- and soft-tissue preservation and regeneration have been utilized in achieving an optimal soft-tissue profile both esthetically and functionally allowing for improved long-term implant stability.
DISCLOSURE
Dr. Leventis is a consultant to Sunstar Suisse SA, Etoy, Switzerland.
About the Authors
Minas D. Leventis, DDS, MS, PhD
Researcher, Department of Oral & Maxillofacial Surgery, Dental School, University of Athens, Greece; Private Practice, Oral Surgery and Implant Dentistry, Athens, Greece
Peter Fairbairn, BDS
Visiting Professor in Periodontology and Implant Dentistry, School of Dentistry, University of Detroit Mercy, Detroit, Michigan; Private Practice, Implant Dentistry, Kensington, London, UK
Robert A. Horowitz, DDS
Departments of Periodontics and Implant Dentistry, Oral Surgery, New York University College of Dentistry, New York, New York; Private Practice, Periodontics and Implant Dentistry, Scarsdale and New York, New York
References
1. Horowitz R, Holtzclaw D, Rosen PS. A review on alveolar ridge preservation following tooth extraction. J Evid Based Dent Pract. 2012;12(3 suppl):149-160.
2. Keith JD Jr, Salama MA. Ridge preservation and augmentation using regenerative materials to enhance implant predictability and esthetics. Compend Contin Educ Dent. 2007;28(11):614-621.
3. Horowitz RA, Rohrer MD, Prasad HS, et al. Enhancing extraction socket therapy with a biphasic calcium sulfate. Compend Contin Educ Dent. 2012;33(6):420-428.
4. Wang HL, Tsao YP. Mineralized bone allograft-plug socket augmentation: rationale and technique. Implant Dent. 2007;16(1):33-41.
5. Harel N, Moses O, Palti A, Ormianer Z. Long-term results of implants immediately placed into extraction sockets grafted with β-tricalcium phosphate: a retrospective study. J Oral Maxillofac Surg. 2013;71(2):e63-e68.
6. Schmidlin PR, Nicholls F, Kruse A, et al. Evaluation of moldable, in situ hardening calcium phosphate bone graft substitutes. Clin Oral Implants Res. 2013;24(2):149-157.
7. Schenk RK. Bone regeneration: Biologic basis. In: Buser D, Dahlin C, Schenk RK, eds. Guided Bone Regeneration in Implant Dentistry. London, UK: Quintessence Publishing; 1995:49-100.
8. Wennström JL, Derks J. Is there a need for keratinized mucosa around implants to maintain health and tissue stability? Clin Oral Implants Res. 2012;23 suppl 6:136-146.
9. Leventis MD, Fairbairn P, Dontas I, et al. Biological response to β-tricalcium phosphate/calcium sulfate synthetic graft material: An experimental study. Implant Dent. 2014;23(1):37-43.
10. Chan HL, Lin GH, Fu JH, Wang HL. Alterations in bone quality after socket preservation with grafting materials: a systematic review. Int J Oral Maxillofac Implants. 2013;28(3):710-720.